-

Precision medicine and complex care environments. Dynamics in market access and reimbursement.
The health technology space has never been that exciting… and challenging at the same time. Gene therapies, microbiome products, digital therapeutics – 2023 was full of exciting market entries from the emerging healthtech field.
-
Starting 2025: Don’t forget to look beyond the JCA
While we are certainly all on lookout for the first EU Joint Clinical Assessments (JCA), there are quite a few other access, financing, pricing and reimbursement news and changes to watch as 2025 starts…
-

Exploring access and reimbursement of digital medical devices and AI-based SaMD at ISPOR Europe 2024
How to approach reimbursement and access of DMDs? DMD reimbursement remains a highly dynamic, where complex access options need to be carefully evaluated, followed and potentially re-evaluated. While access strategy needs to be tailored to each device, some general questions for the DMD field should be considered for each evidence generation plan and go-to-market plan:…
-

Weighing the Costs: Reimbursement of Anti-Obesity Medicines in the US and Europe
With the global burden of obesity continuing to rise, the tension between established reimbursement rules, increased efficacy of new anti-obesity medicines and the need to tackle this population health issue is becoming more pronounced. Reimbursement policies for anti-obesity medications vary significantly by region and are driven by country-specific healthcare priorities, reimbursement policies and budget constraints,…
-

Uncertainties Surrounding the EU Joint Clinical Assessment: What do biotechs need to monitor
With the implementation of the new EU Health Technology Assessment (HTA) Regulation just four months away, uncertainties remain. The most important ones we will look at in this short clip, before exploring strategies for success the next time.
-

Nononsense EU JCA
No-nonsense EU HTA Introducing bit-sized no-nonsense insights about the EU HTA Regulation, Joint Clinical Assessment, Joint Scientific Consultations, etc etc. In a series of posts, there will be Q&A, process overviews, insights about preparedness, uncertainties and unknowns, as well as news as they happen in the continuing implementation phase. Got a question or query to…
-

The dynamics in diagnostic imaging tenders and funding
Reimbursement, access, competition and purchasing are highly dynamic in the diagnostic imaging technology field.
-

Apersy’s 1st anniversary
I started with the vision to create independent market access services that can support health tech companies through the challenges of precision medicine, complex treatment regimens and care approaches that blur the boundaries between device, pharmaceuticals and digital.
-
Announcement of UK IDAP Pilot for medical devices – What does it mean for reimbursement?
The Innovative Devices Access Pathway (IDAP) Pilot is a first step to providing a faster and supported route to regulatory approval, access and adoption of innovative devices. But what precisely does this mean ultimately for access and reimbursement?
-
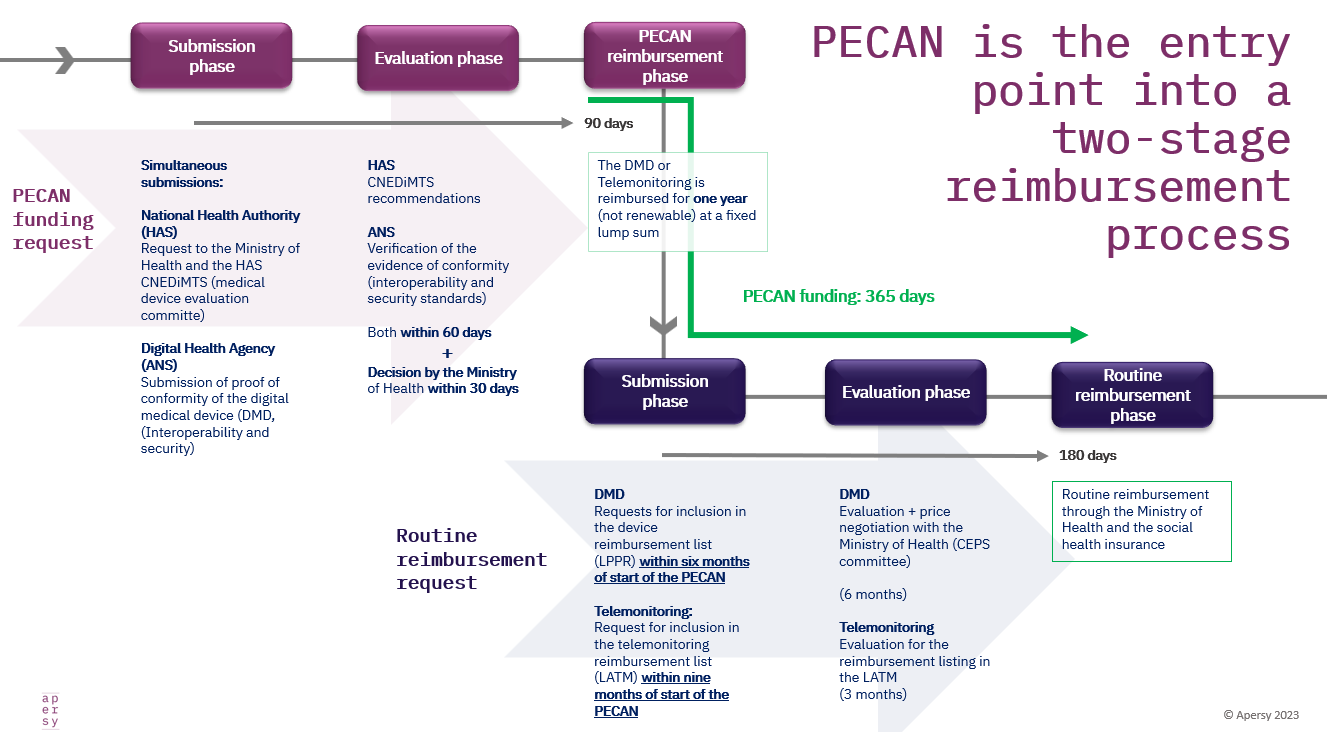
French PECAN reimbursement applications have started… It’s not a DiGA copy-paste
…. the fundamental differences in the French and German access paths for digital medical devices